Medical devices save countless lives each year and can dramatically improve the quality of life of millions of people. However, the development and manufacturing of advanced medical devices is a complex, time-consuming and investment-intensive process.
In a bid to scale up their services, life sciences and healthcare service providers need well-curated technology to market their products and services.

More than the device, the technology used in the backend to curate the information and compile it is important than ever.
Here’s a beginner's guide to the medical device CMS.
What is a Medical Device CMS?
Healthcare professionals who are responsible for technology management require a balanced and effective content management system with possible medical device integration.
A medical device CMS is the one that provides an easy integration matrix between a medical device (hardware) and a data management system (software).
The main purpose of it is to provide a better monitoring of the data collected by the hardware devices while storing it for the future.
Some of the best examples of medical devices would include insulin monitor, fitness tracker bands, heart rate monitoring devices - to detect stroke, sleep tracking devices, among others.
A medical device CMS balances the hardware-software relationship
An ideal medical device CMS address the following:
- Ability to store and forward the acquired data from a hardware device
- Manage and format the data in the required standards
Here's how Drupal is faring in the Healthcare technology trends.
From Pulse to the Report
The dramatic increase in smartphone usage and portable devices have triggered the emergence of stand-alone health and medical devices and software apps.
Wearable sensor-devices play a critical role in monitoring the health of a person. These devices can record real-time information about one's physiological condition and motion activities.
These devices can be identified on the basis of their sensors and accordingly fall in the predefined categories. They are capable of measuring physiological signs such as heart rate, body temperature, blood pressure, respiration rate, electromyogram, electrodermal activity, and in most cases even insulin.
Wearable sensor-devices play a critical role in monitoring the health of a person.
Micro-electro-mechanical system (MEMS) based miniature motion sensors are used to measure activity related to signals from the body movement.
The measured and processed data is then transmitted to the remote healthcare facility (or to the smartphones) over the internet. Security, here, is the major concern since a secured communication channel would safeguard the privacy of (sensitive) personal medical data of an individual.
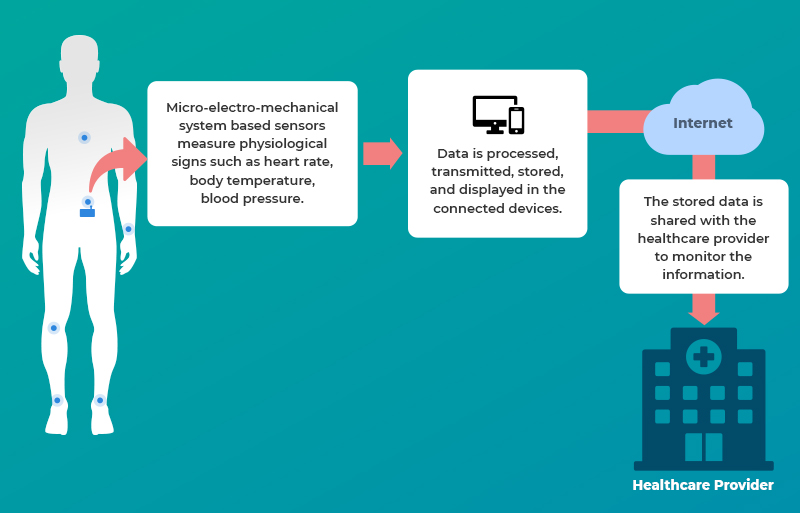
Tips for Medical Device Developers and Organizations
When planning to build a healthcare device, it is important to choose a CMS that gives an accurate representation of data and disseminates the data (in different forms of content) to different platform while adapting to the various screens and sizes too.
The CMS must provide the following features:
- Security: Under the HIPAA Privacy rules, healthcare services providers must ensure automatic log off, encryption and decryption of data, tracking logs that record activity on hardware and software.
The technology at the backend must ensure that the health data can be accessed by limited people and secured under definitive measures. SSL certificate is one of the implementations.
- Remote Access: The information procured from the medical device should be easily accessible to the individual and healthcare provider, who can then monitor the changes. Easy tracking of data is on screen with various elements present while providing the necessary training material in itself.
- User-friendly: Usability is an important aspect of medical device designing. A balance between aesthetics and usability is important for small devices like track bands. Use of flat design, icons, enabling scrolling, 3D look, small text to reduce visual density can give a cleaner look with a minimalistic look.
- Responsive presentation of the data: While medical devices can be of different shapes and screen sizes, the CMS must ensure that the content is presentable in various mobile devices to keep the user interface intact. The flow (dissemination) of content must be consistent flowing on to the screens regardless of the device.
Exploring the Medical Device CMS. Here’s What Healthcare Professionals Need.
As against the common misapprehension, a CMS is responsible not just for the written content but for every piece of information that can be used to create and manage digital documents.
In the above section, the process of the gathering information has been discussed in details and it should come as no surprise how important a CMS is in the process.
Not any CMS can be the choice for a medical device. This list will help you filter out the features of a CMS required from an ideal choice.
-
An Effective Document and Content Management
Establishing a system for mapping and managing the documentation of various users and their records can be a heftier task.
When selecting a CMS for medical devices, healthcare organizations recognize the need to have easy document management solutions.
The CMS must ease the process of generating and maintaining records resulting from the micro-electro-mechanical system while presenting them in different types as per the requirement. As the service provider, it should be able to track, manage, and store information and documents related to the patient’s health.
A medical device CMS needs to be an effective document management system.
Supporting a wide variety of document types, an effective solution would seamlessly integrate all procured health information (scanned charts and data, clinical paperwork, referrals, and more) into the patient’s profile.
Additionally it should provide enough storage to hold all the patients’ data without letting your application or website bend or break.
Before narrowing down to one, ensure that the CMS supports the following content types:
- Text documents
- Graphics
- Web pages
- All image types
- Reports
- Multimedia
-
Multiple Device Support
Assuming that the user must be using one particular device type would mean a major blunder on the user interface. When developing a sophisticated web application compatible with different devices, it is important to ensure the CMS provides provision to elements like HTML5 to build a responsive application.
Adding a rich user experience using various capabilities will ensure various stakeholders (such as physicians and patient) accessing the desired information easily. It should act as a centralised content dissemination platform for serving digital content on screens.
Text format, SMS, e-mail and other alerts on mobile phones can cover the rapidly growing smaller screens generation.
-
A Secure and Privacy of Data
While electronic methods provide increased efficiency and mobility, HIPAA ensures that appropriate steps are taken by the organization to safeguard the information and maintain the integrity and privacy of ePHI.
Medical devices share patient information with other technologies which could compromise patients’ privacy and security if hacked. Holding personal information like name, contact information, patient’s medical information, security can’t be compromised when considering a CMS for medical devices.
Without proper security protocols in place, personal and confidential information could be misused by any person or organization interested in exploiting the data for personal gain.
Under HIPAA, a series of regulatory standards are outlined to ensure the protected health information (PHI) is not disclosed and remain safe under the law.
The CMS must ensure a secure access to the system with data encryption repeatedly happening in the backend. Salting and hashing of the database, strong password policy, session limits, and single sign-on systems are enforced.
Proper data validation can be done to prevents XSS, CSRF, DDOS, and other malicious data entry.
-
Easy integration with Third-Party Application
A perfect product doesn’t exist.
A CMS that would offer you all the features without external support doesn’t exist. Since medical healthcare providing organizations are repeatedly moving towards electronic health records (EHR) to store the medical history and offer a splendid digital experience to the patients.
An ideal CMS here would provide easy integration and smooth workflow after integration without compromising on the security.
Working with a lot of devices and data, there are chances of inconsistency and content duplicacy, in order to avoid these issues in real time, a possible synergy needs to be created between the CMS and the third party tools. CMS compatibility with the third party tools is important.
-
Web Accessibility
To ensure universal healthcare in today's times, governments penalize healthcare facilities not complying with web accessibility guidelines.
People with disability face inequality, violation of dignity, and face injustices in walks of life. As their support system, how can the healthcare organizations which are supposed to act as the pillar of support not enforce web accessibility guidelines on their own website?
From the legal perspective, penalties can risk reputational loss, financial loss, or a class-action lawsuit.
Failing to meet the web accessibility standards, healthcare service providers can leave the disabled patients frustrated and discriminated against.
Everything has shifted online and patients are seeking information online more than they are consulting doctors for real. Web accessibility is more crucial than ever for the healthcare industry as differently abled people jump to receive information online.
Poor contrast, no resize feature, no aural alerts, no keyboard navigation access can leave many visitors with bitterness and confusion. Therefore, it is important for an ideal CMS to provide web accessibility features at its core. If not user experience then under section 508, organizations are compelled to make all web technologies fully usable by people with various disabilities.
The role of the CMS doesn’t end with integrating various devices and process, the more complied with the web accessibility law, the better can healthcare service provider serve quality healthcare. Not only do healthcare organization reach out with an improved brand message but consumer experience as well.
-
A Centralized Content Management System
“A content repository is a database of (digital) content with an associated set of data management, search and access methods allowing various application-independent access to the content with the ability to store and modify content.”
The proliferation of content from a variety of sources can create an enormous challenge. As the unstructured content grows, data loss becomes a hard reality. Healthcare organizations need to look for a flexible approach that supports interoperability with a wide array of popular systems and products.
A CMS that is robust and can be worked as a central content management repository storing a variety of content formats, facilitating read/write capabilities, control access. Information shared through one platform (say a fitband) to be updated in real-time across all the connected systems (EHR System).
As a content repository, it must provide efficient storage to integrate content, easy versioning, import/ export of content, content manager accessibility for all the documents and the content, records retention management system providing the capability of document-imaging.
-
Community-based CMS
Communities help you evolve. Promoting direct communication between service provider and end user, communities can actually boost the product development cycle. Handling a diverse set of stakeholders as a means of increasing quality management and regulatory compliance while reducing risk, the provision of building communities can ease one thing for marketers.
Covering general FAQs, or major technical problems, communities provide the user with a platform to connect with.
Software as a Medical Device
In an August 2016, the Food and Drug Administration (FDA) released a draft guidance document which details it as one of the fastest growing trends in medical devices: Software as a Medical Device, or SaMD.
With “one week workout” and “six-minute cardio” going crazy for the now health conscious millennials medical devices are a big success. To ensure the safety of these devices and the people using them, they need to pass the quality check by the FDA.
Software as a Medical device is a software intended to be used for one or more medical purposes that perform the objective without being part of a hardware medical device, 2014, IMDRF report.
It is a concept proposed by FDA. Prepared with precise guidelines and regulations, FDA assesses the safety and efficacy of new devices that go to the market.
Software intended to be used for one or more medical purposes that perform the objective without being part of a hardware medical device.
SaMD may interface with other physical devices, but it must run on general computing platforms (or mobile devices), and may be used in combination with other medical devices.
Some examples of SaMD devices are:
- Software that allows MRI and other types of medical imaging to be viewed on regular mobile devices
- Software that performs image processing in to detect cancer
- Treatment planning applications that supply information
- Software that regulates an installed medical device, like a pacemaker
- BMI and body fat calculators, and heart rate monitors
An important distinction here is that the software doesn’t meet the definition of SaMD if its intended purpose is to power a hardware medical device.
Accordingly, the categories for software medical devices distinguish software applications across two key dimensions:
- The significance of Information – Devices that are used directly in the treatment or diagnosis of patient illnesses are expected to obtain higher standards of clinical evidence, including obtaining both scientific and analytical validity, as well as assessing clinical performance.
- State of Disease – When a SaMD is used as an intervention for a critical disease, it must be tested more rigorously than if its intended use is in detecting non-serious illnesses.
Conclusion
Being fit is the new fad. Because of which healthcare-medical devices are increasingly becoming popular. At the same time, the importance of the software behind the curtain cannot be denied.
With the rapid development in technologies such as wireless, embedded, nanotechnology and so on it has become possible to develop handy systems and devices.
At OpenSense Labs, we understand that with technology advancing, healthcare software needs to be an all-rounder. They need to be secure, robust and easy to integrate with other technologies and platforms.
Drop a mail at [email protected] or give a shout out on Twitter @OpenSenseLabs to talk about possible synergies.
Subscribe
Related Blogs
What is HTMX & How it Works for Server-Driven Web Interfaces?

“HTML was designed to explain user interactions. HTMX pushes that behavior back where it started, the markup itself." This…
DrupalCon Chicago: Key Product & AI Updates

“The DrupalCon Chicago keynote looks back at Drupal’s 25-year journey while outlining how the platform is evolving. It…
DrupalCamp Delhi Returns After 6 Years: Here’s What to Expect

“After the COVID period, this marks the first time the camp is returning to Delhi. Over the years, the camp and the local…




